Safety & Adverse Event Reports
It is essential that all Principal Investigators are familiar with and comply with the requirements of the Safety Monitoring and Reporting in Clinical Trials Involving Therapeutic Goods (NHMRC, 2016).
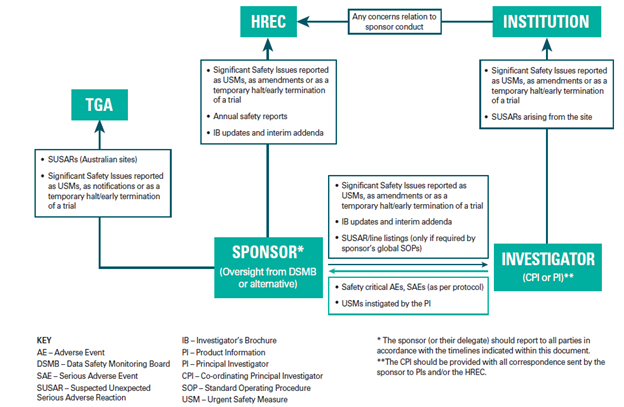
NHMRC Safety Reporting Flowchart
Responsibilities of the Principal Investigator
Investigators should assess all local safety events and should act on any events as clinical care dictates. The role of the investigator with regard to safety reporting is to provide the sponsor with all relevant information so that an appropriate safety analysis can be performed.
The Principal Investigator should:
- Capture, assess and report to the sponsor all Adverse Events (AE) that occur at the site as required and in accordance with the protocol / clinical investigation plan.
- Report to the Sponsor pregnancies that occur while a participant is on a clinical trial as specified in the clinical investigation plan and follow-up any pregnancy until outcome (e.g. birth or spontaneous abortion).
- Report to the Sponsor within 24 hours of becoming aware of the event:
- All Serious Adverse Events (SAEs) or Serious Adverse Device Effects (SADEs) except those that are identified in the protocol as not needing immediate reporting.
- Any occurrences of congenital anomaly/birth defect arising from any pregnancy of a participant (or partner).
- All Urgent Safety Measures (USM) instigated by the site
- Report to the sponsor as specified in the protocol:
- All safety critical events
- Any additional requested information relating to reported deaths
- Report to the institution (Western Health) within 72 hours of becoming aware of the event:
- All Significant Safety Issues (SSIs)
- Suspected Unexpected Serious Adverse Reaction (SUSARs) or Unanticipated Serious Adverse Device Effect (USADEs) arising from the local site.
What to Report?
Investigators should report the following safety information only for participants at Western Health sites to the Western Health Office for Research:
- All SSIs and SUSARs/USADEs details to be reported within 72 hours to [email protected] & the Reviewing HREC.
How to submit Safety Reports
- Complete and submit the Safety Report Sub-Form to the reviewing HREC on ERM
- Note: The Safety Report can only be found under the HREA under Sub-Forms. If you do not have access to the HREA Form for your project, please contact the applicant listed under the HREA and request authorisation to access these forms
- Submit a copy of the ERM Safety Report to WH RGO via email: [email protected]. Provide a copy of the HREC acknowledgment if available.
- Name the file and the subject of the email so that it includes the project number, the type of document and the date it was sent (such as 41234 Safety Report Form 14Nov20). Refer to our mandatory electronic naming convention (download on right hand side links).
- Acknowledgement and/or queries will be emailed to you by the Office for Research in due course.
